- Blog
13/01/2026
Mice, the species most commonly used for scientific purposes, are sentient animals whose welfare depends directly on their living conditions.
All aspects of housing and handling laboratory mice are inherently stressful. This is due to handling, restraint, invasive and painful procedures, unnatural environments, social discomfort, cage cleaning, and more (Bailey, 2018).
In SPF and SOPF facilities (Specific Pathogen Free / Specific and Opportunistic Pathogen Free), sanitary requirements further increase these stress factors:
• During handling, because of the use of biological safety cabinets with air circulation, lighting, and detergent odors;
• In housing rooms, through the use of IVCs (Individually Ventilated Cages) requiring specialised racks that can generate noise and vibration.
This article focuses specifically on the influence of the macro-environment (parameters of the animal housing room) on the welfare of laboratory mice maintained under SPF or SOPF conditions.
A first article described the primary environmental parameters: temperature, humidity, light intensity and photoperiod, noise, and vibrations.
In this second part, the modulatory factors of these primary environmental parameters are discussed: ventilation and housing density.

Role of air handling units (AHU)
SPF and SOPF animal facilities require a controlled environment to maintain high sanitary standards. Air handling units (AHU) allow control of relative pressures between rooms, air exchange rates, as well as humidity and temperature.
Ventilated racks use room air to ventilate cages. Relative pressures between cages and the room are maintained through the air exchange rate in the cages, and the humidity and temperature of the incoming and outgoing airflow are typically displayed for continuous monitoring.
The outgoing air from the rack can be directed either back into the housing room or outside.
Impact on the microenvironment
Room-level ventilation affects cage ventilation as it modulates temperature and humidity (and consequently CO₂ and ammonia levels), which are parameters that can influence animal welfare (see above).
A malfunctioning AHU or incorrect configuration (particularly of relative air pressures) can pose a sanitary risk, potentially compromising the health of housed animals.
Effects on environmental parameters
Housing rooms can contain multiple racks, each holding up to a hundred cages, with each cage housing up to five mice (under TransCure bioServices conditions).
Excessive density in a housing room can lead to changes in environmental parameters:
• Noise: animals are a source of noise through their cage activity and vocalizations (including ultrasounds not perceived by humans); staff-generated noise may also increase in high-density rooms due to increased workload or handling time.
• Vibrations: vibrations from equipment (including racks) will be greater at higher density, as well as vibrations from staff activity.
• Temperature and humidity: if ventilated racks are not connected to external air, increased density will modulate these parameters in the room (and consequently CO₂ and ammonia levels).
• Olfactory compounds: higher density also increases pheromone exchanges (a stressed or suffering mouse can transmit its distress to other mice in the same or neighboring cages), which can affect animal behavior (Lovasz et al., 2020; Sterley et al., 2018).
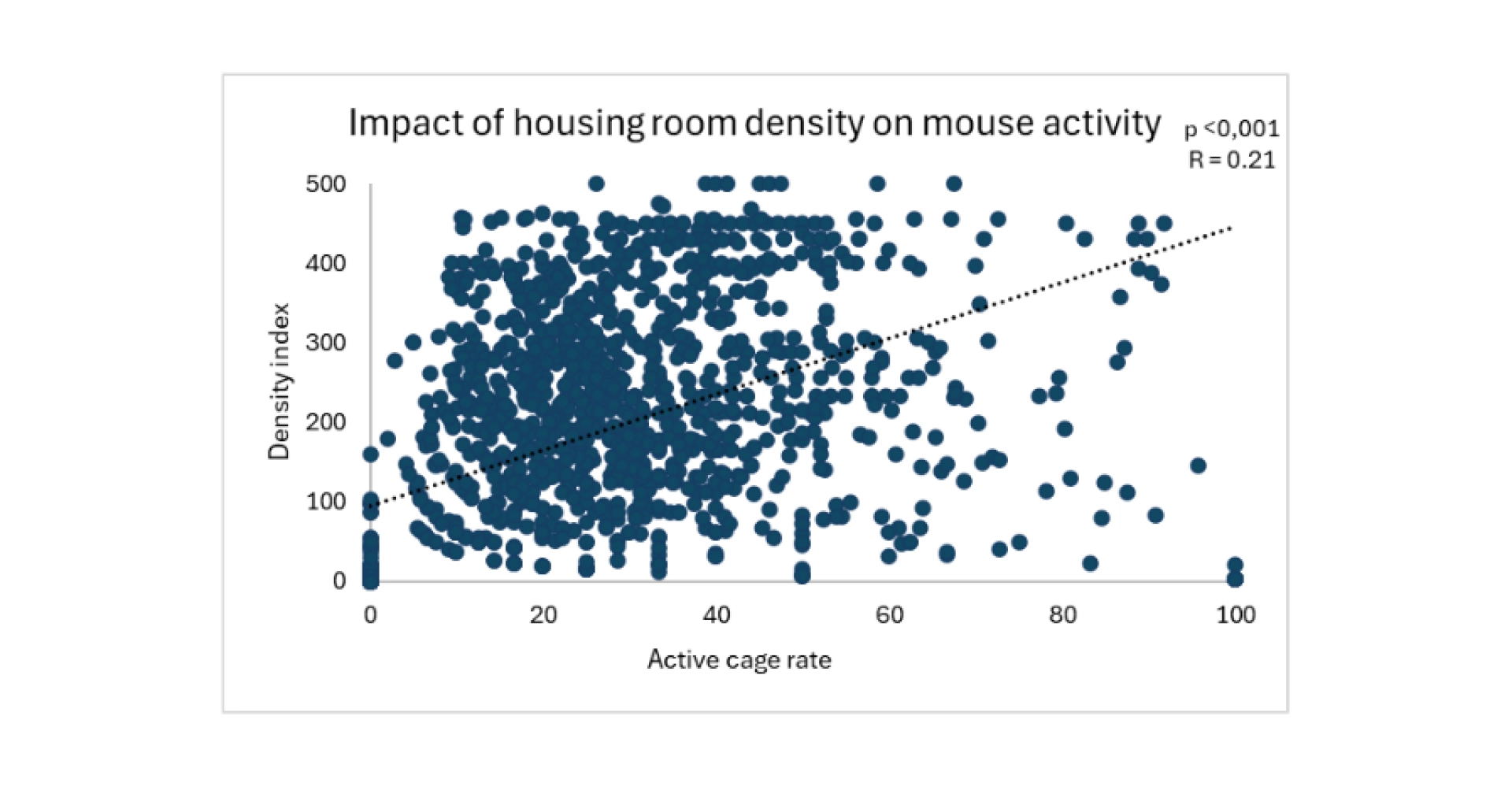
At TransCure bioServices, we focus on maintaining low density in housing rooms, favoring smaller rooms with fewer racks rather than overcrowded large rooms. We have observed a significant positive correlation between a density index (rack occupancy percentage*number of racks) and mouse activity (measured visually using an internally developed method) (see figure below).
Figure 2: Impact of a density index (rack occupancy rate × number of racks in the housing room) on mouse activity. As the data were non-parametric (Shapiro tests), a Spearman correlation was used
Many environmental parameters can impact animal welfare. They are all interconnected, providing multiple levers for action in case of difficulty.
Ventilation in housing rooms controls temperature and humidity, which directly affect these same parameters in mouse cages. Ammonia and CO₂ levels are directly influenced, particularly by humidity.
Light conditions affect mouse activity, which in turn generates noise (a parameter related to density).
To improve the welfare of animals used for scientific purposes, and therefore the quality of scientific results, it is essential to understand all of these parameters and their interdependence. For more information on housing conditions and animal welfare practices at TransCure bioServices, you can consult our other blog articles.
Bailey, J. (2018). Does the stress of laboratory life and experimentation on animals adversely affect research data? A critical review. In ATLA Alternatives to Laboratory Animals (Vol. 46, Issue 5, pp. 291–305). FRAME. https://doi.org/10.1177/026119291804600501
Barabino, S., Rolando, M., Chen, L., & Dana, M. R. (2007). Exposure to a dry environment induces strain-specific responses in mice. Experimental Eye Research, 84(5), 973–977. https://doi.org/10.1016/J.EXER.2007.02.003
Briese, V., Fanghanel J., & Gasow, H. (1984). Effect of pure sound and vibration on the embryonic development of the mouse. Zentralblatt Fur Gynakologie, 106(6), 379–388.
Canadian Council on Animal Care. (2019). CCAC guidelines: Mice (CCAC, Ed.; CCAC). Canadian Council of Animal Care. http://www.ccac.caACKNOWLEDGEMENTS
Carman, R. A., Quimby, F. W., & Glickman, G. M. (2007). The effect of vibration on pregnant laboratory mice. INTER-NOISE and NOISE-CON Congress and Conference Proceedings, 1722–1731.
European Community Council. (1986). Directive 86/609/CEE.
European parliament and of the council of 22 September 2010 on the protection of animals used for scientific purposes. (2010). Directive 2010/63/EU.
Fawcett, A. (2012). Guidelines for the Housing of Mice in Scientific Institutions.
Fischer, A. W., Cannon, B., & Nedergaard, J. (2018). Optimal housing temperatures for mice to mimic the thermal environment of humans: An experimental study. Molecular Metabolism, 7, 161–170. https://doi.org/10.1016/j.molmet.2017.10.009
Ganeshan, K., & Chawla, A. (2017). Warming the mouse to model human diseases. Nature Reviews Endocrinology 2017 13:8, 13(8), 458–465. https://doi.org/10.1038/nrendo.2017.48
Garner, J. P., Gaskill, B. N., Gordon, C. J., Pajor, E. A., Lucas, J. R., & Davis, J. K. (2018). Impact of nesting material on mouse body temperature and physiology Author’s personal copy Impact of nesting material on mouse body temperature and physiology. https://doi.org/10.1016/j.physbeh.2012.12.018
Garner, J. P., Gaskill, B. N., Rohr, S. A., Pajor, E. A., & Lucas, J. R. (2011). Working with what you’ve got: Changes in thermal preference and behavior in mice with or without nesting material Author’s personal copy Working with what you’ve got: Changes in thermal preference and behavior in mice with or without nesting material. Article in Journal of Thermal Biology. https://doi.org/10.1016/j.jtherbio.2011.02.004
Gordon, C. J. (2017). The mouse thermoregulatory system: Its impact on translating biomedical data to humans. Physiology & Behavior, 179, 55–66. https://doi.org/10.1016/J.PHYSBEH.2017.05.026
Hessler, J. R., & Leary, S. L. (2002). Design and Management of Animal Facilities. Laboratory Animal Medicine, 909–953. https://doi.org/10.1016/B978-012263951-7/50024-7
Hofstetter, J. R., Hofstetter, A. R., Hughes, A. M., & Mayeda, A. R. (2005). Intermittent long-wavelength red light increases the period of daily locomotor activity in mice. Journal of Circadian Rhythms 2005 3:1, 3(1), 1–8. https://doi.org/10.1186/1740-3391-3-8
Hylander, B. L., & Repasky, E. A. (2016). Thermoneutrality, Mice, and Cancer: A Heated Opinion. Trends in Cancer, 2(4), 166–175. https://doi.org/10.1016/j.trecan.2016.03.005
James, C. M., Olejniczak, S. H., & Repasky, E. A. (2022). How murine models of human disease and immunity are influenced by housing temperature and mild thermal stress. https://doi.org/10.1080/23328940.2022.2093561
Lovasz, R. M., Marks, D. L., Chan, B. K., & Saunders, K. E. (2020). Effects on Mouse Food Consumption after Exposure to Bedding from Sick Mice or Healthy Mice. Journal of the American Association for Laboratory Animal Science, 59(6), 687–694. https://doi.org/10.30802/AALAS-JAALAS-19-000154
Norton, J. N., Kinard, W. L., & Reynolds, R. P. (2011). Comparative Vibration Levels Perceived Among Species in a Laboratory Animal Facility. Journal of the American Association for Laboratory Animals, 50(5), 653–659.
Peterson, E. A. (1980). Noise and laboratory animals. Laboratory Animal Science, 30(2), 422–439.
Recordati, C., Basta, S. M., Benedetti, L., Baldin, F., Capillo, M., Scanziani, E., & Gobbi, A. (2015). Pathologic and Environmental Studies Provide New Pathogenetic Insights Into Ringtail of Laboratory Mice. Veterinary Pathology, 52(4), 700–711. https://doi.org/10.1177/0300985814556191
Repasky, E. A., Hylander, B. L., & Mohammadpour, H. (2024). Temperature matters: the potential impact of thermoregulatory mechanisms in brain–body physiology. Genes & Development, 38(17–20), 817–819. https://doi.org/10.1101/GAD.352294.124
Reynolds, R. P., Li, Y., Garner, A., & Norton, J. N. (2018). Vibration in mice: A review of comparative effects and use in translational research. In Animal Models and Experimental Medicine (Vol. 1, Issue 2, pp. 116–124). John Wiley and Sons Inc. https://doi.org/10.1002/ame2.12024
Rosenbaum, M. D., VandeWoude, S., Volckens, J., & Johnson, T. (2010). Disparities in Ammonia, Temperature, Humidity, and Airborne Particulate Matter between the Micro-and Macro environments of Mice in Individually Ventilated Caging. Journal of the American Association for Laboratory Animal Science, 49(2), 177–183. https://www.researchgate.net/publication/42767874_Disparities_in_Ammonia_Temperature_Humidity_and_Airborne_Particulate_Matter_between_the_Micro-and_Macro_environments_of_Mice_in_Individually_Ventilated_Caging
Silverman, J., Bays, D. W., & Baker, S. P. (2009). Ammonia and carbon dioxide concentrations in disposable and reusable static mouse cages. Lab Animal, 38(1).
Sterley, T. L., Baimoukhametova, D., Füzesi, T., Zurek, A. A., Daviu, N., Rasiah, N. P., Rosenegger, D., & Bains, J. S. (2018). Social transmission and buffering of synaptic changes after stress. Nature Neuroscience, 21(3), 393–403. https://doi.org/10.1038/s41593-017-0044-6
Turner, J. G. (2020). Noise and Vibration in the Vivarium: Recommendations for Developing a Measurement Plan. Journal of the American Association for Laboratory Animal Science, 59(6). https://doi.org/10.30802/AALAS-JAALAS-19-000131
Zhang, Z., Wang, H.-J., Wang, D.-R., Qu, W.-M., & Huang, Z.-L. (2017). Red light at intensities above 10lx alters sleep-wake behavior in mice. https://doi.org/10.1038/lsa.2016.231